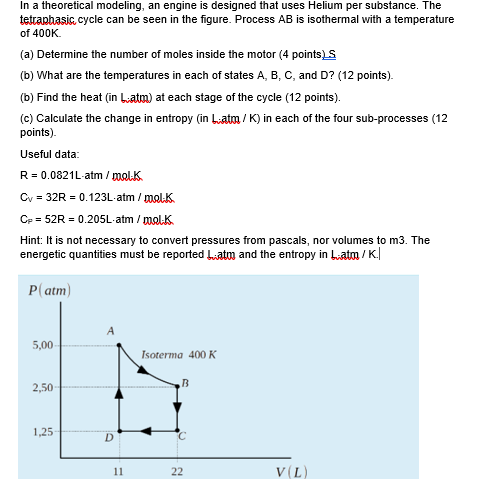
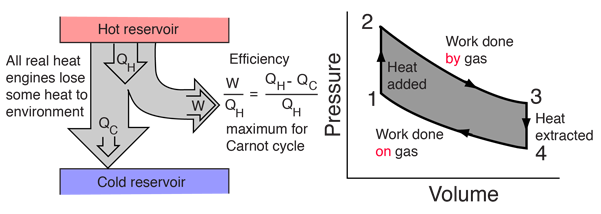
15.14 | Calculate the net work output of a heat engine following path ABCDA in the figure below. - YouTube

SOLVED:The heat engine shown in HGURE P19.60 uses 2.0 mol of a monatomic gas as the working substance. a. Determine T1, T2, and T3 b. Make a table that shows ΔEth, Ws,
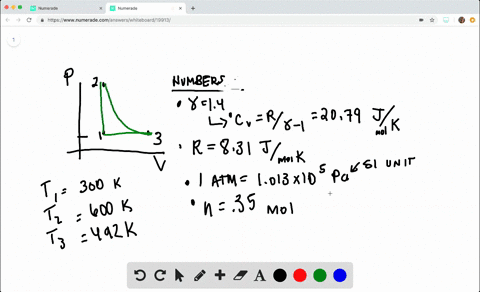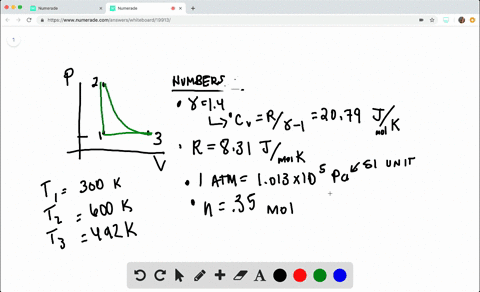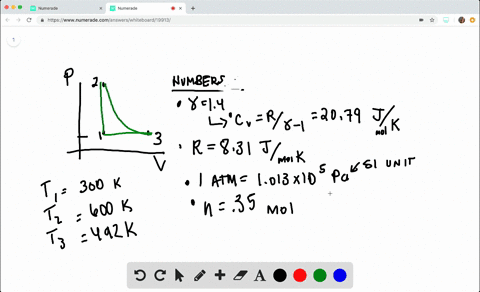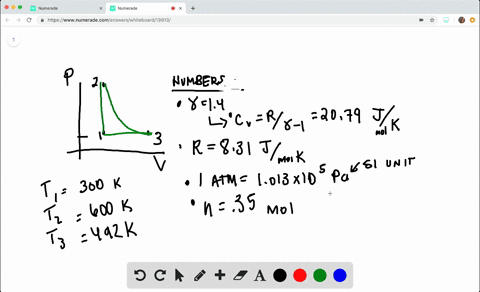
SOLVED:A heat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of Fig. P20.36. Process 1→2 is at constant volume, process 2→3 is adiabatic, and

SOLVED: A heat engine using 140 mg of helium as the working substance follows the cycle shown in the figure igure 1) Figure 1 of 1 (atm) Isotherm (cm ) Vnnx 1000

SOLVED:A heat engine uses 100 . mg of helium gas and follows the cycle shown in the figure. a) Determine the pressure, volume, and temperature of the gas at points 1 ,
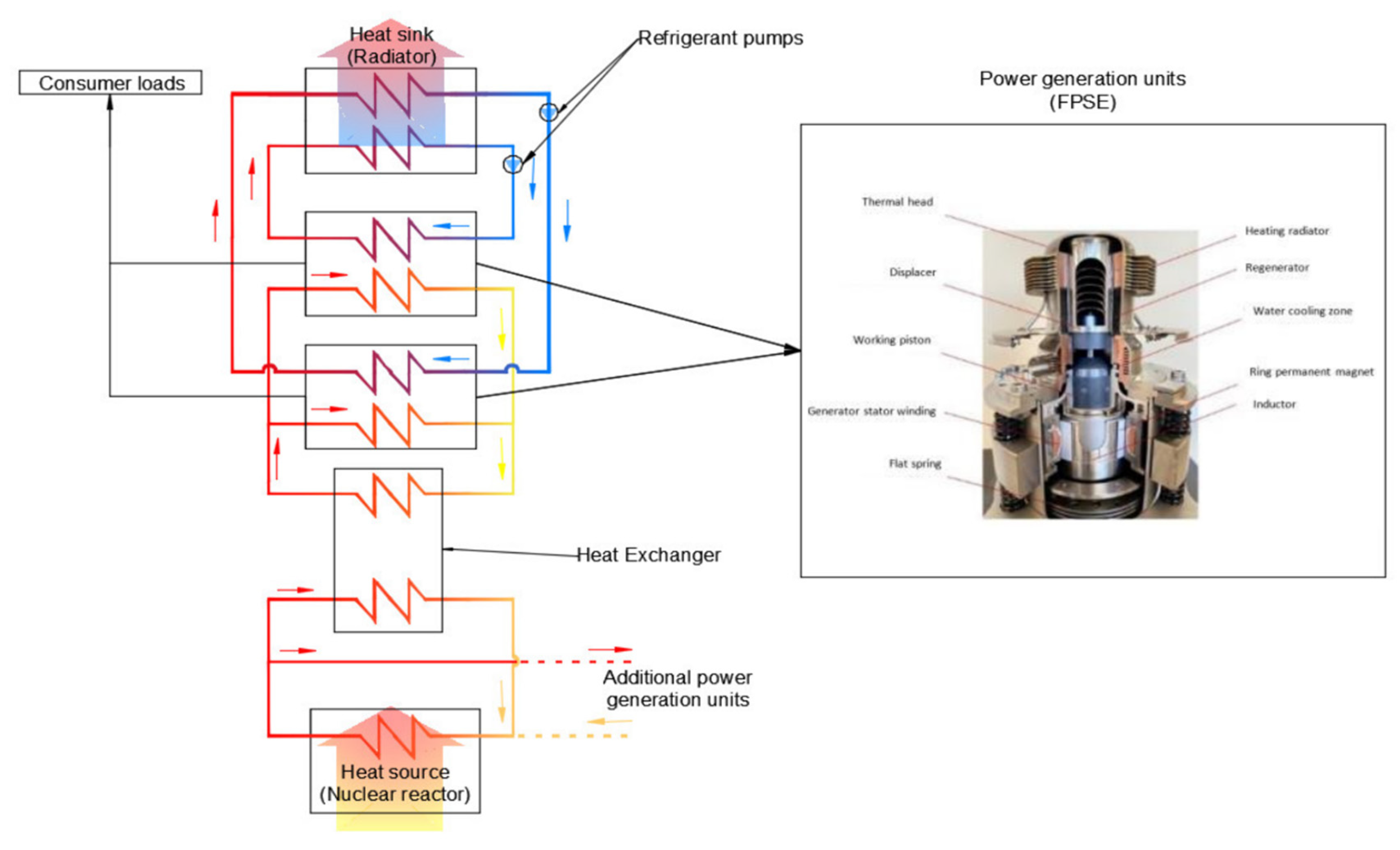
Symmetry | Free Full-Text | An Enhanced Calculation Method of the Heat Rejection System of a Free-Piston Stirling Engine (FPSE) Operating on the Moon
Week 14 Homework Assignment - n moles of helium are initially at a temperature of T0 and occupy a - Studocu

Calculate the difference between the two specific heats of Helium gas per gram. Given that molecular weight of He = 4, J = 4.18 joule/cal and R = 8.31 J mole^-1//K^-1?