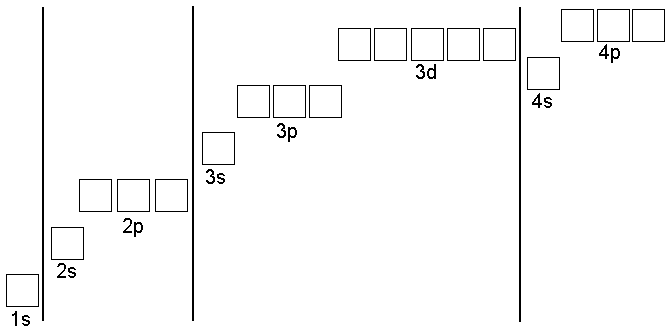
![If each orbital can hold a maximum of 3 electrons, the number of elements in the 4th bb"[period of the]" periodic table (long form) is? | Socratic If each orbital can hold a maximum of 3 electrons, the number of elements in the 4th bb"[period of the]" periodic table (long form) is? | Socratic](https://useruploads.socratic.org/Nyy0D0VSOKyNIw578PCP_electrons_in_boxes.jpg)
If each orbital can hold a maximum of 3 electrons, the number of elements in the 4th bb"[period of the]" periodic table (long form) is? | Socratic

An 'electrons-in-box' or energy level representation of the electronic... | Download Scientific Diagram

Write the full electron configuration, the orbital box diagram, and the noble gas shorthand configuration for the element with the following atomic number: Z=38 | Homework.Study.com
Electron Configurations, how to write out the s p d f electronic arrangements of atoms ions Periodic Table oxidation states using orbital notation GCE A level revision notes
Write the full electron configuration, the orbital box diagram, and the noble gas shorthand configuration for the element with the following atomic number: Z=14 | Homework.Study.com
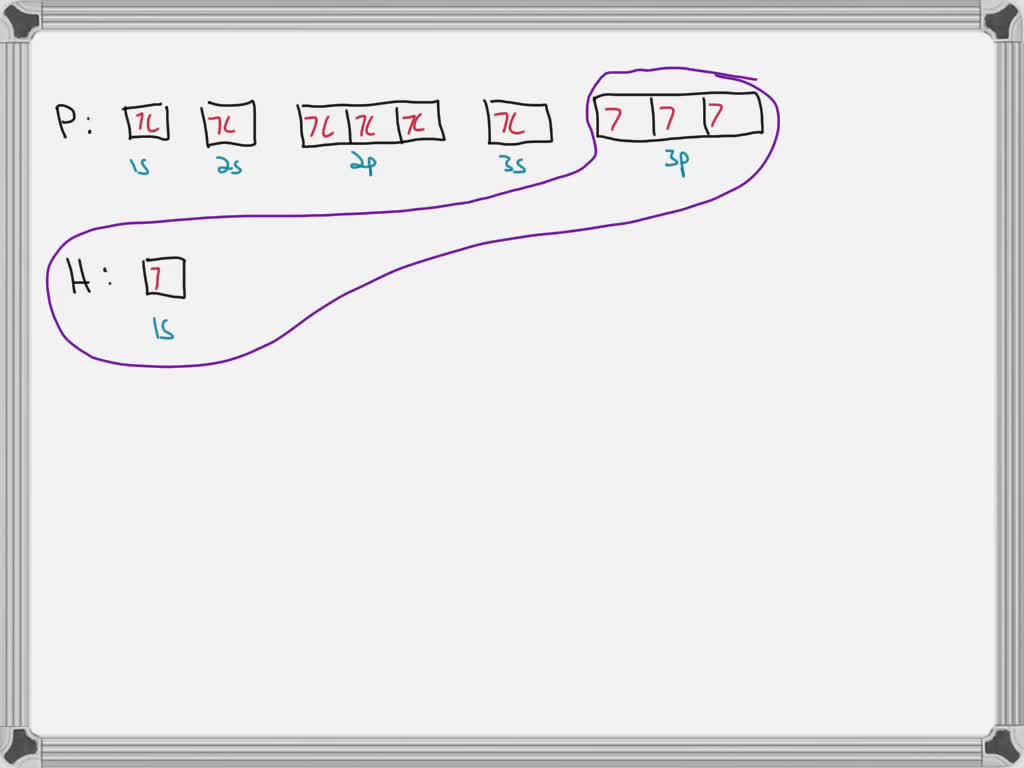
SOLVED: Draw orbital diagrams (boxes with arrows in them) to represent the electron configurations-without hybridization-for all the atoms in PH3. Circle the electrons involved in bonding. Draw a three-dimensional sketch of the

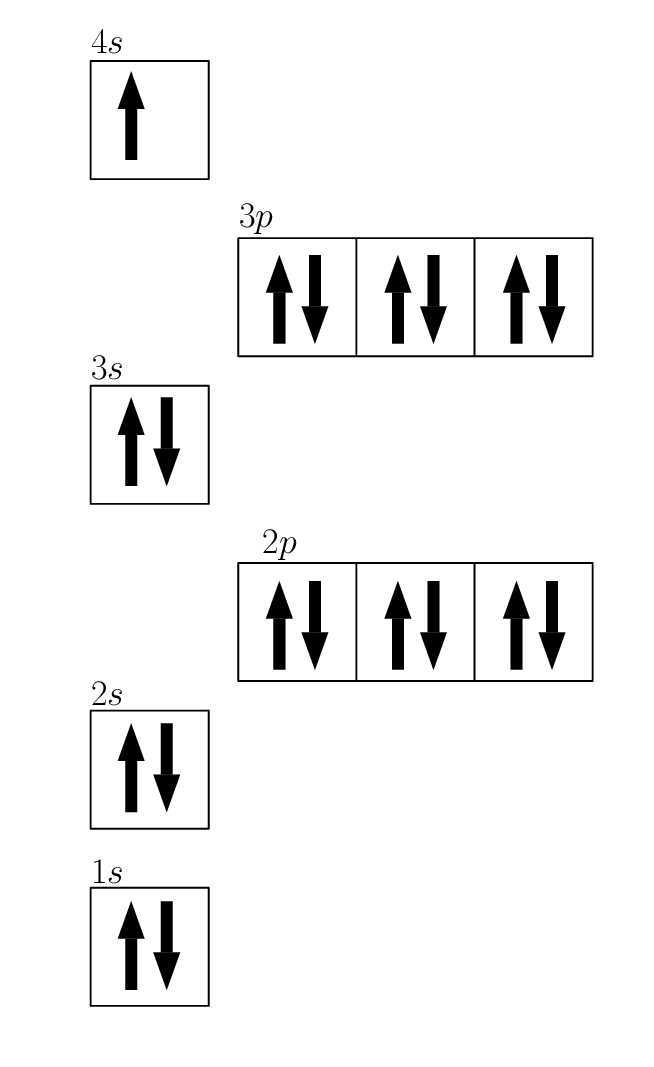
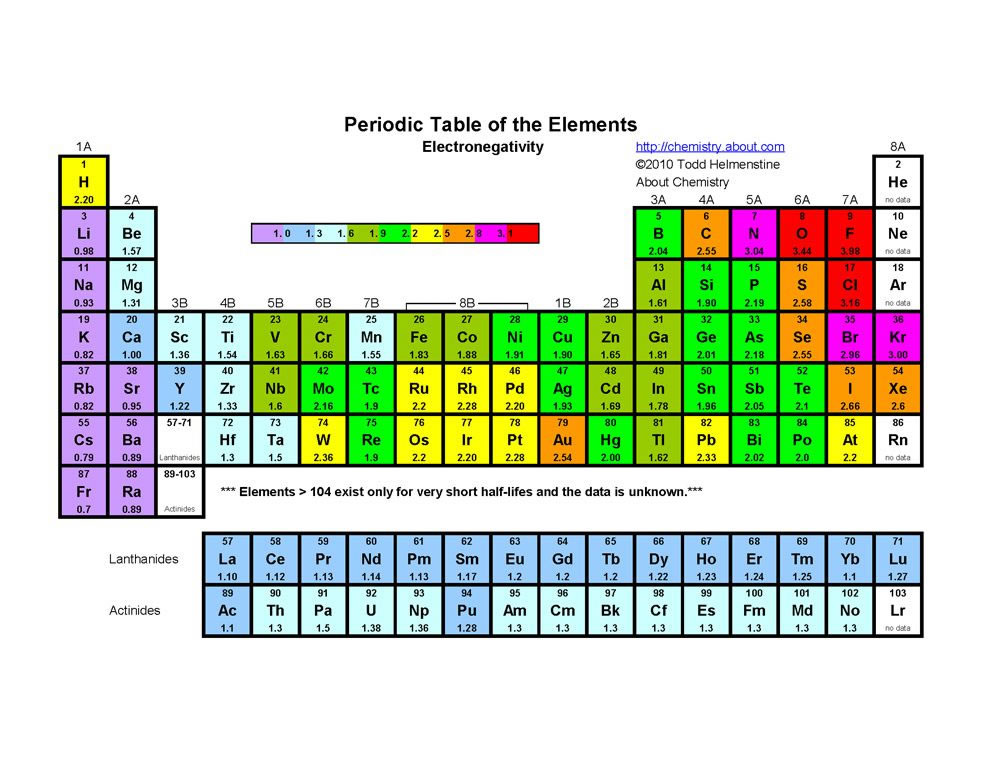





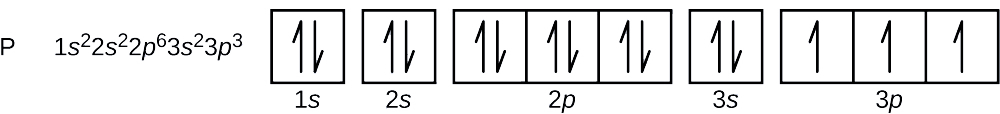


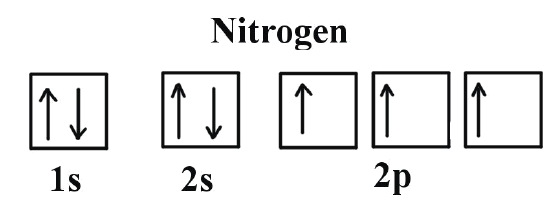
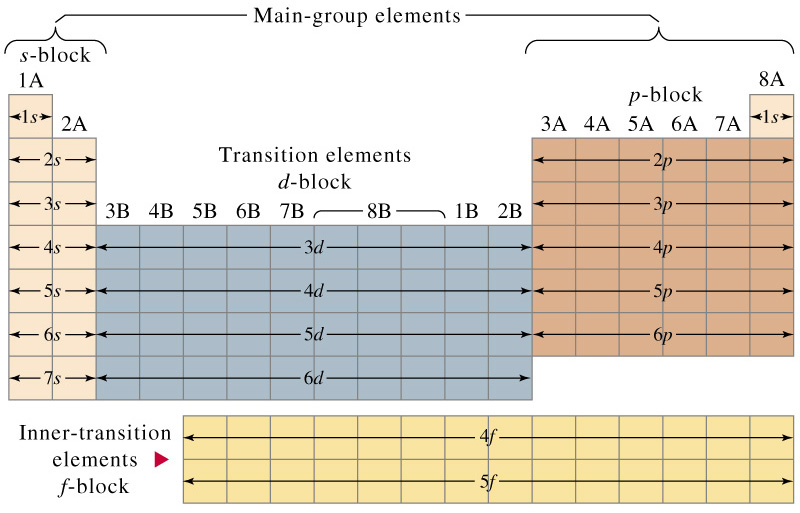
![How to draw Electron-in-box diagrams Electronic Configurations? [GCE A Level Chemistry] - YouTube How to draw Electron-in-box diagrams Electronic Configurations? [GCE A Level Chemistry] - YouTube](https://i.ytimg.com/vi/JzmL6kktPRY/maxresdefault.jpg)